Clinical trial packaging
- No minimum batch size, from Phase I through III
- In-house design and prototyping in 5 to 10 days
- GMP-certified, GDP-compliant, FMD serialization ready







Protocol changes should not derail your packaging timeline
Clinical supply managers know the reality: protocols shift, dosing regimens change mid-study, and patient numbers fluctuate between phases. Every amendment means revised packaging specifications, often on tight deadlines. When your packaging supplier cannot adapt quickly, delays cascade through your entire clinical supply chain.
Ecobliss operates as a single team of 40 specialists with both manual and automated packaging lines. This means we scale up or down without procurement cycles or retooling delays. A protocol amendment on Monday can be a revised packaging run by the end of the week, not the end of the quarter.

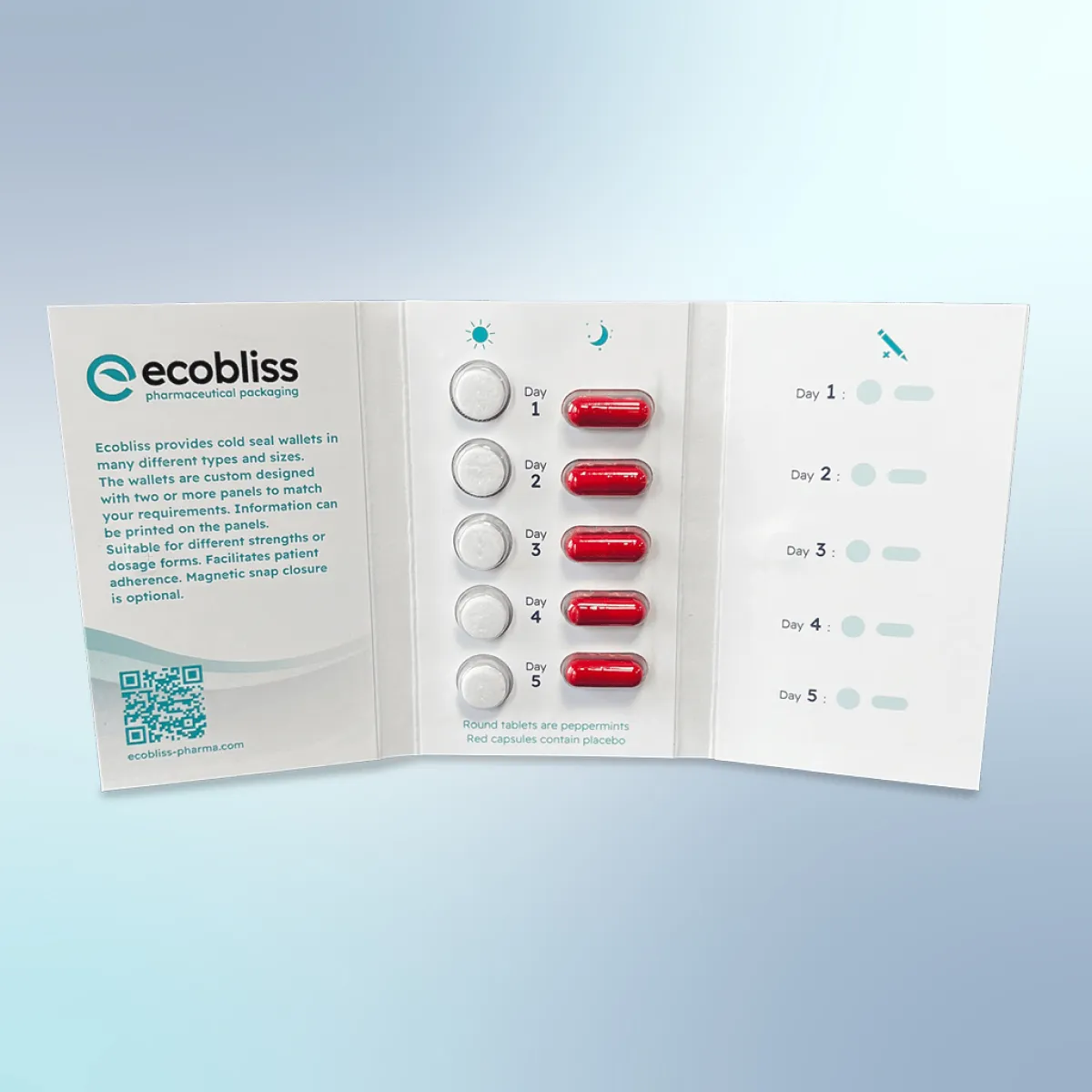
Cold seal technology built for sensitive compounds
Our cold seal wallet uses pressure-only sealing with a water-based, solvent-free adhesive. No heat is applied at any point in the process. For temperature-sensitive compounds, this eliminates a significant risk factor. The wallet format also integrates printed dosing panels directly into the packaging, reducing the need for separate patient information inserts.
Beyond cold seal, we provide FMD-compliant serialization across all formats, GDP-compliant warehousing including secure storage for Schedule I and II controlled substances, and full traceability from raw material to finished pack. 90% of our pharmaceutical clients choose cold seal after evaluating their options.

Your partner from first batch to commercial launch
Most clinical packaging suppliers treat each phase as a separate project. You re-qualify, re-validate, and re-brief at every transition. Ecobliss works differently. One team manages your project across all clinical phases and into commercial production. The knowledge built during Phase I carries through to market launch without handoffs or information loss.
With 30 years of pharmaceutical packaging experience and clients including Organon, Lundbeck, and Novartis, we understand that clinical packaging is not a one-off job. It is the foundation for your commercial supply chain. We design every clinical pack with the commercial transition already in mind.
Reduce your supplier coordination burden
Managing multiple vendors for design, prototyping, packaging, serialization, and warehousing consumes project management hours that should go toward your trial. Ecobliss consolidates these functions under one roof in Echt, the Netherlands. Design, prototyping, GMP packaging, FMD serialization, and GDP-compliant storage, all from a single qualified supplier.
This integration shortens your supply chain, reduces audit burden, and gives you one point of accountability. When timelines compress or specifications change, there are no vendor-to-vendor handoffs to manage. One brief, one team, one delivery.
How we deliver your clinical trial packaging
Briefing and feasibility
We review your compound requirements, phase, batch sizes, and regulatory needs. You receive a feasibility assessment and packaging concept within days, not weeks.
Design and prototyping
Our in-house team creates packaging designs and functional prototypes in 5 to 10 days. You evaluate physical samples before committing to production.
GMP production and serialization
Packaging runs on our manual or automated lines under full GMP conditions. FMD-compliant serialization is applied as required. Batch sizes flex to match your trial enrollment.
Storage and distribution
Finished packs are held in our GDP-compliant warehouse, including controlled substance security where needed. We coordinate dispatch to your clinical sites or logistics partner.
Ready to simplify your clinical supply chain?
Talk to our team about your upcoming trial. We will assess your requirements and show you how a single packaging partner reduces complexity from Phase I through commercial launch.
Frequently asked questions about clinical trial packaging
What is your minimum batch size for clinical trials?
We have no fixed minimum batch size. Whether you need 50 packs for a Phase I study or thousands for a Phase III trial, we scale production to match your actual enrollment numbers.
How quickly can you produce prototypes?
Our in-house design and prototyping team delivers functional samples within 5 to 10 working days. This includes packaging structure, printed components, and dosing panel layouts.
Can you handle controlled substances?
Yes. We hold GDP-compliant warehousing with security provisions for Schedule I and II controlled substances. All handling follows documented procedures with full traceability.
What does cold seal mean for my compound?
Cold seal uses pressure-only sealing with a water-based, solvent-free adhesive. No heat is applied during the sealing process. This makes it suitable for temperature-sensitive compounds where heat exposure during packaging would be a risk.
Do you support the transition from clinical to commercial packaging?
Yes. One team manages your project from Phase I through commercial launch. The packaging design, processes, and documentation developed during clinical phases transfer directly to commercial production without re-qualification.
What serialization capabilities do you offer?
We provide full FMD-compliant serialization, including unique identifiers, tamper-evident features, and data reporting to the relevant national verification systems.
Where are you located and which markets do you serve?
Ecobliss is based in Echt, the Netherlands. We serve pharmaceutical companies across Europe and internationally, with GDP-compliant distribution to clinical sites worldwide.




